By Dr. Erin Madigan-Fleck NMD, LE, LEI
For millions of years, the epidermis has effectively separated life from an outer world of environmental elements, microbes, and toxins. Scientists have long theorized about the epidermis's evolutionary development and the differences between the human integumentary system and that of our potential primitive ancestors.
Anthropologist Nina Jablonski has elucidated that principles of historical morphology can be used to reconstruct the evolution of human skin by studying the phylogeny of organisms, thus providing insight into the evolutionary alterations in one of life’s most essential organs.
Human skin, over millions of years has been subject to fundamental changes both in structure and function as compared to gastrointestinal and respiratory linings, and the skin's immune system has evolved into a complex defense mechanism. The skin is a major component of the body's innate immune system, with inherent capabilities and defensive functions. Archeological studies worldwide have shown how changes in the environment have impacted the skin, and how its epithelial linings developed gene-encoded antimicrobial peptides (AMPs) to defend against environmental threats and microbes.
The discovery of Cationic Intrinsically Disordered Antimicrobial Peptides (CIDAMPs) can be traced back to the beginning of life and continues to be present in the skin today. Keratinocytes, neutrophils, and macrophages collaborate to regulate innate immunity, and the interaction between keratinocytes and myeloid cells, known as keratinocyte-myeloid synergy (KMS), plays a crucial role in our skin's immune defense. [1,2]
Epidermal cells transition through a complex continuum of development, commencing from undifferentiated cells in the basal layer, differentiating into secretory cells, and finally forming the stratum corneum. Epidermal functions provide protective and defensive roles during the formation of the epidermal layer and the stratum corneum, defining the final stages of epidermal differentiation.
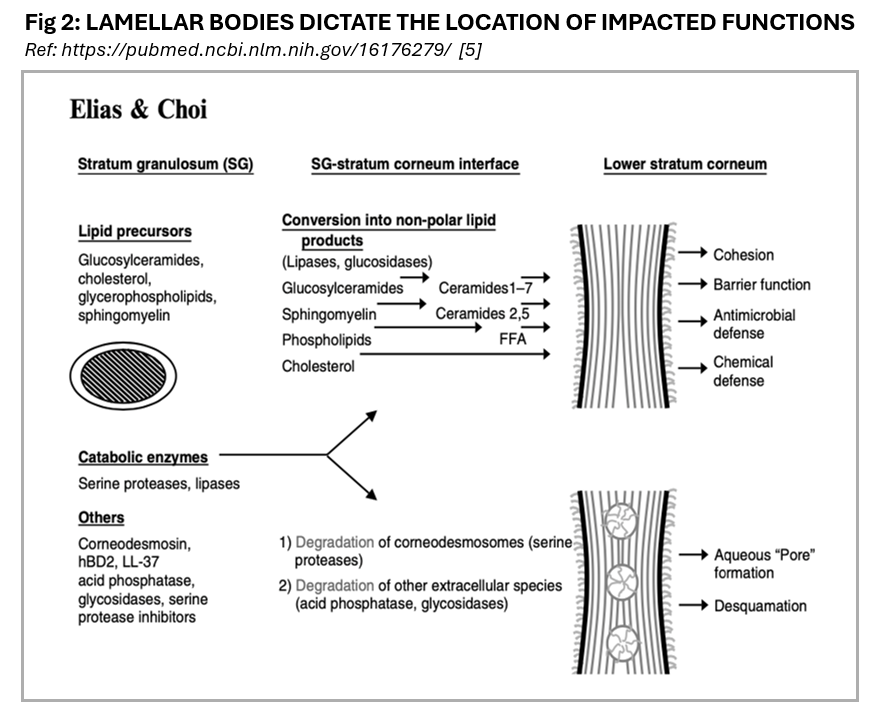
Essential barrier lipids are created in the endoplasmic reticulum, and the Golgi apparatus is secreted and released into a complex system of lamellar bodies that influence the SC's functions. These lipids include glucosyl ceramides - which transform into ceramides, cholesterol, cholesterol esters, and long-chain fatty acids.
Ceramides are regarded as a complex group of sphingolipids that contain derivatives of sphingosine bases, a variety of fatty acids, and cholesterol, and play a crucial role in supporting the barrier properties of the epidermis. Collectively, these lipids constitute the intercellular lipid domain and are a major constituent of the lamellar sheets present in the intercellular spaces of the stratum corneum.
The lipids begin with a partially granular and partially lamellar appearance before being released into the space between the living secretory cells and the cornified cells. Once in this intercellular space, the lipids undergo a transformation and form laterally extended, stacked sheets.
This transformation is completed at the third to fifth layer of cornified cells, and the stacked sheets are observed between the cornified cells throughout the stratum corneum.
It is important to note that most skin disorders with a weakened barrier function will show a decrease in total ceramide content, along with variations in the ceramide patterns (SC). [3]
Defend and Protect
The formation of the epidermal permeability barrier involves delivering the contents of lamellar bodies (LB), also known as Odland bodies (OB), to the interstitial spaces of the stratum corneum. Lamellar bodies (LBs) contain a mixture of polar lipids and a family of hydrolytic enzymes necessary for processing the secreted polar lipids into more hydrophobic products that play a role in barrier function. The lamellar structures are approximately 200-300 nanometers in size and are in the upper spinous and granular cell layers.
Additionally, they secrete lipid-hydrolyzing enzymes, proteolytic enzymes, structural proteins, enzyme inhibitors, and antimicrobial peptides (AMPs).
Antimicrobial enzymes AMPs offer a complex system for expression and protection, increasing responses to microbial infection. Cytokines activate sensory neurons and may include AMPS, including beta-defensins. The process of skin inflammation is mainly influenced by the interaction between pro- and anti-inflammatory mediators, including prostaglandins, eicosanoids, leukotrienes, histamines, and cytokines, which are produced and released from keratinocytes to help regulate the skin's immune responses.
Cathelicidins are unique AMPs that protect the skin via direct antimicrobial activity and the activation of immune responses, initiating the pathway for cytokine release, inflammation, angiogenesis, and re-epithelialization.
There are distinctive hallmarks regarding cathelicidin dysfunction that include suppression in atopic dermatitis and abnormally processed peptides that lead to inflammation in rosacea, and psoriasis. LBs also contain phospholipids, glucosylceramides, sphingomyelin, cholesterol, and proteases such as cathepsin D and kallikrein, and the protein, corneodesmosins. Recent research suggests that LBs also play a role in the skin's immune response by containing AMP beta 2 defensins, which provide effective activity against certain types of bacteria and candida.
The AMP glycoprotein corneodesmosin, which is produced by the human gene CNDS, is also found in corneodesmosomes in the epidermis. AMPs are produced and released from keratinocytes during stress or damage to stimulate immune cells (mast cells), which release pro-inflammatory cytokines [4,5,6]
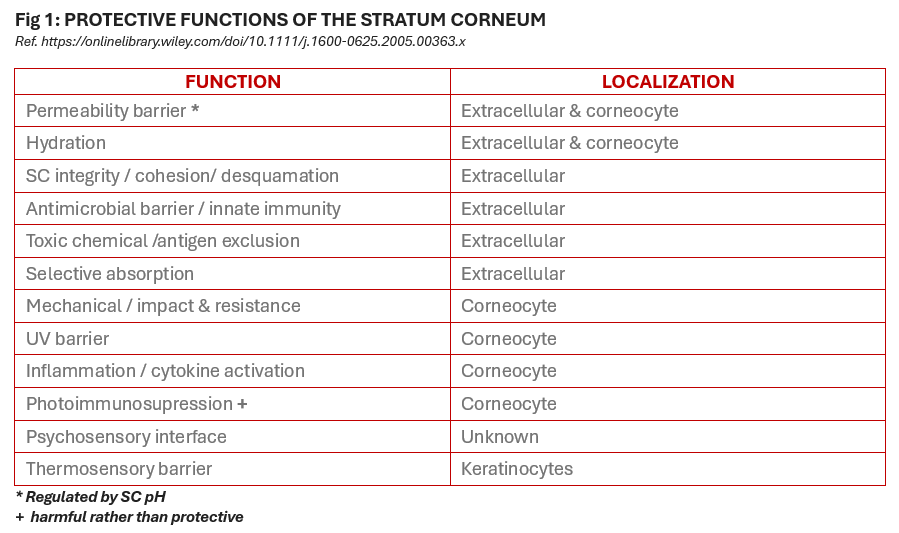
Defensive Functions of The Stratum Corneum SC Are United in Their Protective Capabilities:
• The relationship between permeability and antimicrobial barriers
• The filaggrin pathway influences defensive functions
• Barrier mechanisms repair inflammation while stimulating and supplying permeability
• Elevations of SC pH alter multiple defense functions
• Alterations in the mechanical barrier influence permeability barrier homeostasis
• Oxidative stress generates corneodesmosome proteins to be more susceptible to proteolysis
Capturing Calcium
The distribution of calcium in the skin allows for dynamic changes in calcium ions, facilitating essential signaling processes and various cellular functions in keratinocytes. These functions include differentiation, the formation of the epidermal barrier, and epidermal homeostasis. Evidence suggests that both the release of Ca2+ from intracellular and extracellular sources is important for regulating epidermal structures, functions, and the conversion of pro-filaggrin to filaggrin. Calcium also regulates the transcription of all genes encoding keratinocyte differentiation-specific proteins.
Activator protein-1 (AP-1) transcription factors are present in many keratinocyte-specific genes, including transglutaminase, loricrin, involucrin, pro-filaggrin, and other keratins to control the transcription of various differentiation markers. Studies suggest that the accumulation of calcium ions within the cell's endoplasmic reticulum (ER) contributes to the skin's calcium gradient. Most of the calcium ions measured in the epidermis are derived from intracellular stores in the ER, and the depletion of calcium ions is an important signal for terminal differentiation and maintaining epidermal barrier homeostasis.
The calcium gradient is determined by the relative concentrations of calcium ions in the extracellular and intracellular spaces. Extracellular calcium plays a significant role in forming the skin's calcium gradient and acts as a signal for repairing the skin barrier when it is disrupted. The absence of the normal calcium gradient outside the cells triggers the secretion of lipid barrier components and promotes barrier repair.
Acute disruption of the skin barrier leads to an immediate decrease in extracellular calcium ions in the epidermis, particularly in the upper stratum granulosum, and causes the normal epidermal calcium gradient to be lost. This gradient is formed through various changes and mechanisms in the calcium ion levels when the skin barrier is disrupted. Calcium levels in the upper epidermis gradually recover over 6-24 hours as the barrier is restored. [8,9]
New Horizons
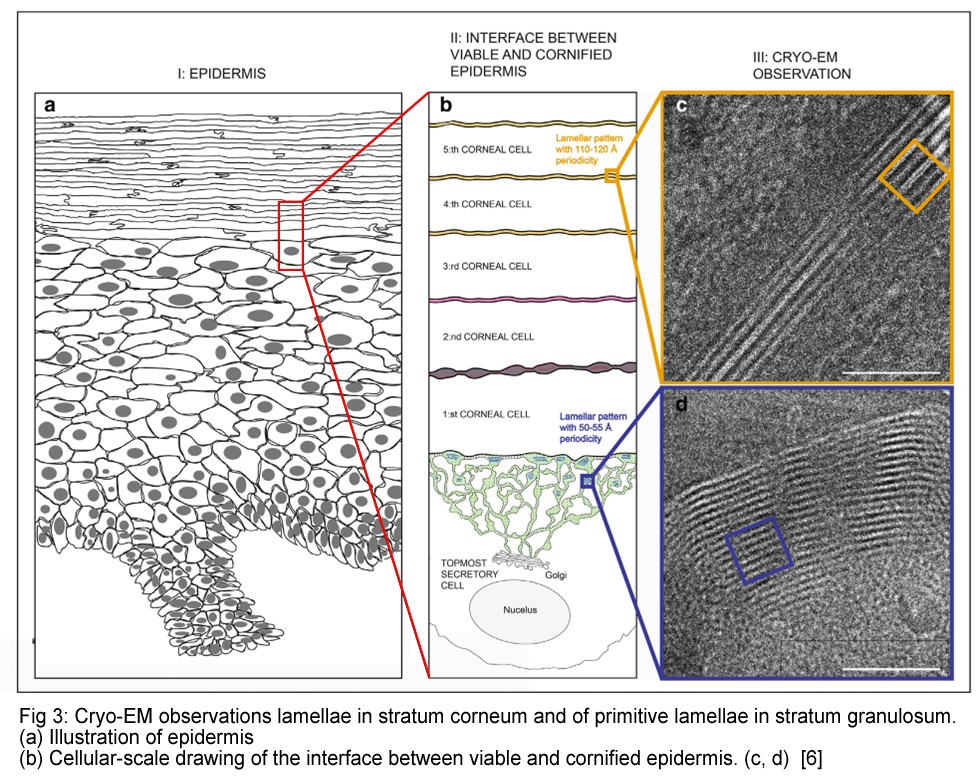
In 2001, Prof. Lars Norlén and his research team at the Karolinska Institute proposed an evolutionary model for the structure and function of the skin barrier. Their research was prompted by the investigation into the potential for improved distribution of transdermal drug delivery into the skin to reduce the side effects of oral administration.
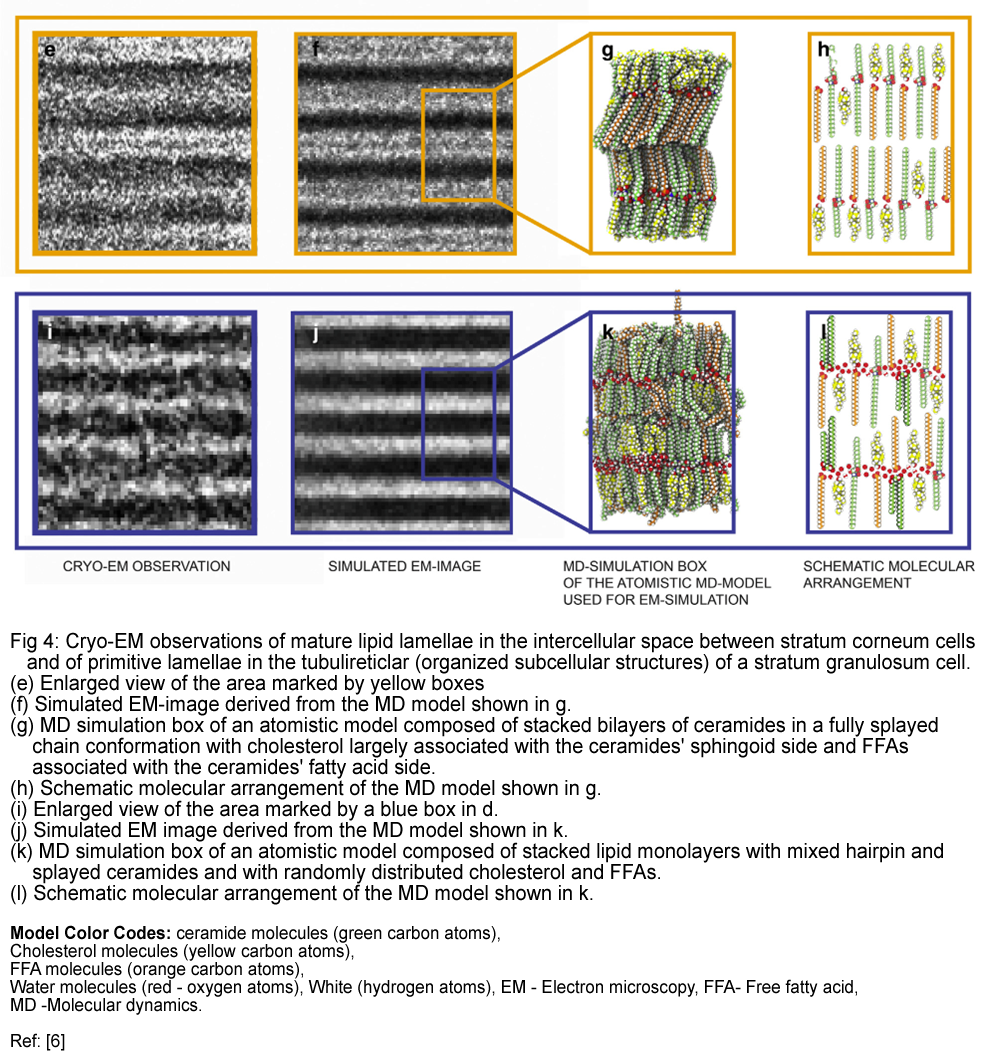
The research involved a combination of cryo-electron microscopy, molecular dynamics modeling, and electron microscopy simulation to analyze the formation of the skin's lamellae. Prof. Norlén’s study revealed two cryo-electron microscopy patterns representing intermediate stages in the maturation process of the lamellae: a single-band pattern and a two-band pattern, which may be derived from the structures of the lamellar lipids.
This unique lipid structure allows the cells in the outer layer of the skin to move against each other, providing flexibility and contributing to the skin's mechanical strength. The study suggests that the outermost layer of the skin contains a single, coherent lamellar gel phase in the intercellular lipid barrier.
This structure primarily resides in the lower half of the stratum corneum, has minimal phase boundaries, and is stabilized by a specific lipid composition and distribution of lipid chain lengths in the intercellular space. This model may explain the measured water permeability of the stratum corneum, the lipid composition of the stratum corneum intercellular spaces, and the absence of swelling of the stratum corneum intercellular lipid matrix upon hydration. [10,11]
Microbiome Alterations and Skin Aging
The human microbiome plays a crucial role in aging as it contains most of the genes expressed in the human body. However, specific signs of aging and skin quality, such as texture, hydration, pH, and sebum levels, may not accurately reflect a person's chronological age.
A 2024 research study has disclosed some relevant insight into the variations of microbiome taxa species and potential microbial features correlated with aging signs, including the epidermis. The microbiota is thought to be stable throughout adulthood; however, microbial communities shift, creating an increase in diversity and skin markers such as sebum quality and dryness due to a reduction of C acnes.
Skin aging characteristics influence immune function and sweat and sebum secretions, resulting in significant alterations in pH and lipid composition. These alterations impact the microbiome and amino acid metabolism pathways and amino acid degradation across various age groups.
The skin microbiome has been found to be correlated with clinical parameters such as UV spots, dark spots, and skin hydration in previous studies. However, it is not always easy to ascertain whether these patterns are the result of the aging process itself or the physiological manifestations of aging.
This recent study presented research that identified taxa that appear to be more related to signs of aging and skin quality parameters than to chronological age. Corneometry metrics were applied to study samples from the cheek skin, as this area is crucial in determining age-related anomalies.
The findings provided further insights into the associations between the skin microbiome, especially specific groups of S. epidermidis or C. acnes, and the biophysical characteristics of skin aging, such as collagen quality and quantity. It was discovered that taxa that correlated with crows’ feet and wrinkles include mainly environmental bacteria such as Kaistella and other taxa from the phylum Actinomycetes (Brevibacterium and Microbacterium).
While some of these types of bacteria can be found on healthy skin, a high abundance of these bacteria on the skin has previously been linked to skin issues caused by air pollution and skin inflammation. Microbacterium was previously found on the skin of a population exposed to high levels of polycyclic aromatic hydrocarbon pollutants. Brevibacterium was reported to be prevalent in skin samples of individuals with psoriatic lesions and senile pruritus.
Several of the bacteria associated with increased crow's feet and wrinkles in the study have been previously identified in the context of skin microbiome dysbiosis linked to various skin inflammatory conditions. This groundbreaking research identifies specific microbial characteristics linked to common signs of aging, as well as skin barrier function measured by TEWL, and skin hydration as indicated by the corneometer. [12]
Resources used in this article:
- https://dl0.creation.com/articles/p149/c14993/j35_3_85-90.pdf
- https://onlinelibrary.wiley.com/doi/pdf/10.1111/exd.14482
- https://www.ajicjournal.org/article/S0196-6553%2806%2900950-3/pdf
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4144227/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2688147/
- https://onlinelibrary.wiley.com/doi/epdf/10.1111/j.1600-0625.2005.00363.x
- https://www.jidonline.org/article/S0022-202X(20)32190-4/fulltext
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5929942/
- https://www.bioscmed.com/index.php/bsm
- https://www.jidonline.org/article/S0022-202X(20)32190-4/fulltext
- https://ki.se/en/research/research-areas-centres-and-networks/research-groups/basic-molecular-structure-of-human-skin-lars-norlens-research#tab-publications
- https://www.frontiersin.org/journals/aging/articles/10.3389/fragi.2023.1304705/full?utm_source=FNTF&utm_medium=EMLX&utm_campaign=PRD_FEOPS_20170000_ARTICLE












